INSIGHTS
SMART events
SMART news
SMART Life Science
SMART Career
SMART Corporate Citizenship and Social Responsibility

Article Series Continuous Improvement in a QMS – the Fil Rouge that Connects all Quality Processes:: 2.1 The Role of the Business Process Owner Within a QMS (Part 1)
PART 1: The Importance and Complexity of the Business Process Owner Role In recent years, the pharmaceutical...

Avoid FDA Warning Letters in the Context of Computerized System Validation: A Short Guide by KVALITO
Photo by Eduardo Dutra on PexelsElevating Compliance in the Life Sciences: Navigating Computerized System Validation...

Article Series Continuous Improvement in a QMS – the Fil Rouge that Connects all Quality Processes::Continuous Improvement within a QMS
Have you ever thought about what is the ‘fil rouge’ that connects all regulations applicable to the pharmaceutical...
Good Documentation Practices in Regulated Environments
"Proper documentation is the only witness of what you have done: in its absence, you did nothing; if it is poor, you...
Meet Dimitrij Gvozdev
Meet Dimitrij Gvozdev We would like you to meet Dimitrij Gvozdev, who is passionate about working with KVALITO and the...
Digitalization of Pathology – A New Approach
Development and Validation of Digital Pathology Digital pathology means the use of technology to accelerate and...
Risk-Based Computerized System Validation (CSV) and Computer Software Assurance (CSA) – Old Wine in a New Bottle?
“Discovering the unexpected is more important than confirming the known.” —George Box, Stuart Hunter, and William G....
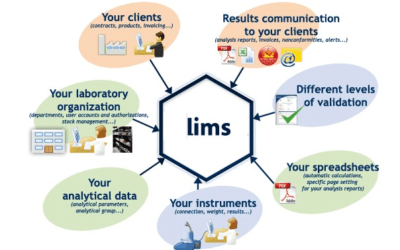
LIMS Implementation: Success Factors from a Project Manager Perspective
"Change before you have to." —Jack Welch A Laboratory Information Management System (LIMS) is software that allows...
Sterilization Processes for Medical Devices
What kind of sterilization methods can be used for a medical device? There are different methods...
CDRH Validation vs. CSV
“A goal without a method is nonsense.” — W. Edwards Deming What is CDRH Validation? In the USA, the FDA’s Centre for...
How to Overcome Validation Costs
“It is thrifty to prepare today for the wants of tomorrow” — Aesop, Greek author (620 BC - 560 BC) Validation...
Medical Devices; US and Chinese legislation
US and Chinese legislation US legislation What is the framework of US Legislation? Figure 1: Framework of FDA...